Reference: https://nba.uth.tmc.edu/neuroscience/toc.htm
- Secrete ADH (Vasopressin)
- Secrete Oxytocin
- Growth Hormone-Releasing Hormone
- Stimulate Thirst
- Hypothalamus-Pituitary-Thyroid Axis
- Temperature Regulation
- Secrete Gonadotrophin
Vasopressin (AVP) is synthesized in magnocellular neurons (Supraoptic nuclei); the distal axons of these neurons project to the posterior pituitary, from which AVP is released into the circulation. Argine vasopressin and oxytocin are stored in Herring bodies at the axon terminals in the posterior pituitary where they are released into the blood stream. These neurons are sensitive to changes in extracellular osmolality. When ECF osmolality increases, these cells shrink and release ADH from the posterior pituitary.
A network of central “osmoreceptor” neurons, which includes the AVP-expressing magnocellular neurons themselves, sense circulating osmolality via nonselective, stretch-activated cation channels.
These osmoreceptor neurons are activated or inhibited by modest increases and decreases in circulating osmolality, respectively; activation leads to AVP release and thirst.
AVP secretion is stimulated as systemic osmolality increases above a threshold level of ~285 mOsm/kg, above which there is a linear relationship between osmolality and circulating AVP.
Thirst and thus water ingestion are also activated at ~285 mOsm/kg, beyond which there is an equivalent linear increase in the perceived intensity of thirst as a function of circulating osmolality.
Effects of ADH
1. On kidney - ADH markedly increases water reabsorption in medullary collecting tubules, which tends to reduce plasma osmolality back to normal.
Conversely, a decrease in extracellular osmolality causes osmoreceptors to swell and suppresses the release of ADH. Decreased ADH secretion allows a water diuresis, which tends to increase osmolality to normal. Peak diuresis occurs once circulating ADH is metabolized (90-120 min). With complete suppression of ADH secretion, the kidneys can excrete up to 10-20 L of water per day.
[Damage to the anterior hypothalamus blocks the production of ADH, resulting in diabetes insipidus, which is characterized by rapid water loss from the kidneys.1]
[Nonosmotic Release of Antidiuretic Hormone
The carotid baroreceptors and probably atrial stretch receptors can also stimulate ADH release following a 5-10% decrease in blood volume. Other nonosmotic stimuli include pain, emotional stress, and hypoxia.]
Oxytocin hormone produce in the Paraventricular nucleus ,at the end of pregnancy, is produced in large amount and stimulates labor contraction of the uterus
These axons (likely, but dendrites have not been ruled out) have collaterals that innervate oxytocin receptors in the nucleus accumbens.
The peripheral hormonal and behavioral brain effects of oxytocin are thought to be coordinated through its common release through these collaterals.[12]Oxytocin is also made by some neurons in the paraventricular nucleus that project to other parts of the brain and to the spinal cord.[111] Depending on the species, oxytocin receptor-expressing cells are located in other areas, including the amygdala and bed nucleus of the stria terminalis.
In the pituitary gland, oxytocin is packaged in large, dense-core vesicles, where it is bound to neurophysin I as shown in the inset of the figure; neurophysin is a large peptide fragment of the larger precursor protein molecule from which oxytocin is derived byenzymatic cleavage.
Secretion of oxytocin from the neurosecretory nerve endings is regulated by the electrical activity of the oxytocin cells in the hypothalamus. These cells generate action potentials that propagate down axons to the nerve endings in the pituitary; the endings contain large numbers of oxytocin-containing vesicles, which are released by exocytosis when the nerve terminals are depolarised.
growth hormone-releasing hormone
Osmoreceptors in the lateral preoptic area of the hypothalamus are very sensitive to changes in extracellular osmolality. Activation of these neurons by increases in ECF osmolality induces thirst and causes the individual to drink water.
Conversely, hypoosmolality suppresses thirst.
Thirst is the major defense mechanism against hyperosmolality and hypernatremia, because it is the only mechanism that increases water intake.
The hypothalamic thermoregulatory center balances excess heat production from metabolic activity in muscle and liver with heat dissipation from the skin and lungs to maintain a normal body temperature of 36.8° ± 0.4°C (98.2° ± 0.7°F), with diurnal variation (lower in a.m., higher in p.m.).
The hypothalamus maintains core body temperature within very narrow range.
- At one end is the threshold for sweating and vasodilation with heat dissipation from the skin and lungs..
- At the other is the threshold for vasoconstriction and shivering (metabolic activity in muscle and the liver)
The human body generates enough heat, even at rest, to raise the body temperature by 1°C every hour.
A normal body temperature is maintained despite environmental variations because the hypothalamic thermoregulatory center balances the excess heat production derived from metabolic activity in muscle (shivering) and the liver
The system is effective such that the daily variation in body temperature is only ±0.6°C (2).
Neurons in both the preoptic anterior hypothalamus and the posterior hypothalamus receive two kinds of signals:
- one from peripheral nerves that transmit information from warmth/cold receptors in the skin
- the other from the temperature of the blood bathing the region.
These two types of signals are integrated by the thermoregulatory center of the hypothalamus to maintain normal temperature.
Increasing core temperature a fraction of a degree induces sweating and vasodilation, whereas a minimally reduced core temperature triggers vasoconstriction and shivering.1
About 90% of the excess heat generated by metabolism is dissipated through the skin, with the remainder being lost in exhaled gas.
Heat exchange between the body and its surroundings is accomplished in several ways:
Under normal conditions, radiation accounts for about 60% of the heat loss from the human body (3).
According to studies of healthy individuals 18–40 years of age, the mean oral temperature is 36.8° ± 0.4°C (98.2° ± 0.7°F), with low levels at 6 a.m. and higher levels at 4–6 p.m.
The maximal normal oral temperature is 37.2°C (98.9°F) at 6 a.m. and 37.7°C (99.9°F) at 4 p.m.; these values define the 99th percentile for healthy individuals. In light of these studies, an a.m. temperature of >37.2°C (>98.9°F) or a p.m. temperature of >37.7°C (>99.9°F) would define a fever. The normal daily temperature variation is typically 0.5°C (0.9°F). However, in some individuals recovering from a febrile illness, this daily variation can be as great as 1.0°C. During a febrile illness, the diurnal variation is usually maintained, but at higher, febrile levels. The daily temperature variation appears to be fixed in early childhood; in contrast, elderly individuals can exhibit a reduced ability to develop fever, with only a modest fever even in severe infections.
Rectal temperatures are generally 0.4°C (0.7°F) higher than oral readings. The lower oral readings are probably attributable to mouth breathing, which is a factor in patients with respiratory infections and rapid breathing. Lower-esophageal temperatures closely reflect core temperature. Tympanic membrane thermometers measure radiant heat from the tympanic membrane and nearby ear canal and display that absolute value (unadjusted mode) or a value automatically calculated from the absolute reading on the basis of nomograms relating the radiant temperature measured to actual core temperatures obtained in clinical studies (adjusted mode). These measurements, although convenient, may be more variable than directly determined oral or rectal values. Studies in adults show that readings are lower with unadjusted-mode than with adjusted-mode tympanic membrane thermometers and that unadjusted-mode tympanic membrane values are 0.8°C (1.6°F) lower than rectal temperatures.
In women who menstruate, the a.m. temperature is generally lower in the 2 weeks before ovulation; it then rises by ∼0.6°C (1°F) with ovulation and remains at that level until menses occur. Body temperature can be elevated in the postprandial state. Pregnancy and endocrinologic dysfunction also affect body temperature.
In a neutral temperature environment, the human metabolic rate produces more heat than is necessary to maintain the core body temperature in the range of 36.5–37.5°C (97.7–99.5°F).
These cytokines stimulate the immune responses of T and B cells, macrophages, and polymorphonuclear leukocytes. They appear to act via a common mechanism involving the induction of prostaglandin synthesis. Feedback inhibitory responses are mediated by adrenocorticotropic hormone, arginine vasopressin, serotonin, dopamine, and other homeostatic mechanisms, thus emphasizing the orchestrated nature of fever production and response to infection. These thermoregulatory mechanisms rarely allow fevers to exceed 41° C (106° F). Temperatures exceeding 41° C are often due to a drug-induced imbalance in these mechanisms and may cause direct cellular damage.
The hypothalamus contains temperature-sensitive neurons that have receptors for pro-inflammatory and anti-inflammatory cytokines, which are continuously balanced to maintain a homeothermic set point. When body temperature becomes elevated, cutaneous vasodilation and sweating occur, and people may reduce activity and seek a cooler environment. In contrast, low body temperature is increased by shivering, piloerection, cutaneous vasoconstriction, adding clothes, and seeking a warmer environment. In a febrile illness, symptoms may be due to the underlying disease or to the fever itself. Malaise is the rule, and many febrile patients experience myalgia secondary to the muscle contractions used to generate temperature elevation. Although it was once thought that the back and thigh pain related to rigors suggests bacteremia, any febrile stimulus can produce such symptoms. The chill associated with rigors may be related to the surface vasoconstriction that accompanies the increase in core temperature.
Bacteria release cell wall products such as peptidoglycans and endotoxin, as well as enterotoxins, which bind to Toll-like receptors (TLRs) on phagocytes (neutrophils, macrophages) and endothelial cells. As a result, pyrogenic cytokines such as interleukin (IL)-1 and IL-6 or tumor necrosis factor (TNF)-α are released into the circulation and bind to cytokine receptors in the hypothalamic organum vasculosum of lamina terminalis (OVLT). Bacterial products may also directly bind to TLRs on the OVLT. Activation of TLRs and cytokine receptors induces cyclooxygenase 2 (COX2), which leads to the production of prostaglandin E2 (PGE2) and elevates cyclic adenosine monophosphate (cAMP) in the brain. This, in turn, triggers neurons in the thermoregulatory center to raise the hypothalamic thermostatic set point. In addition, neuronal signals to the cortex prompt behavioral changes to conserve heat (e.g., posturing, adding clothing). The hypothalamus also triggers sympathetic peripheral efferent nerves that constrict peripheral blood vessels and conserve central heat until hypothalamic PGE2 levels fall.
Among the broad range of conditions that cause fever are infections, malignancies, autoimmune diseases, metabolic diseases, chronic inflammatory conditions, medications (including immunizations), CNS abnormalities, and exposure to excessive environmental heat. (How do these produced pyrogens??)
The hypothalamic set point increases, causing peripheral vasoconstriction (i.e., heat conservation). [The patient feels cold as a result of blood shunting to the internal organs.ref?] Mechanisms of heat production (e.g., shivering, increased hepatic thermogenesis) help to raise the body temperature to the new set point. Increases in peripheral prostaglandin E2 account for the nonspecific myalgias and arthralgias that often accompany fever. When the set point is lowered again by resolution or treatment of fever, processes of heat loss (e.g., peripheral vasodilation and sweating) commence.
Anesthetic agents inhibit central thermoregulation by interfering with these hypothalamic reflex responses. For example,isoflurane produces a dose-dependent decrease in the threshold temperature that triggers vasoconstriction (3°C decrease for each percent of inhaled isoflurane). Both general and regional anesthetics increase the interthreshold range, albeit by different mechanisms. Spinal and epidural anesthetics, like general anesthetics, lead to hypothermia by causing vasodilation and internal redistribution of heat. The accompanying thermoregulatory impairment from regional anesthetics that allows continued heat loss is likely due to altered perception by the hypothalamus of temperature in the anesthetized dermatomes rather than a central drug effect, as with general anesthetics.
Moderate activity increases thermogenesis and results in a transiently increased temperature until heat-dissipating processes are engaged. Each 1° F increase in temperature results in a 7% increase in the basal metabolic rate. Vaporization from the lungs and skin accounts for a third of basal body heat loss and for as much as all heat loss at ambient dry temperatures above 36° C. The elderly have a decrease in basal metabolism, as well as blunted responses to thermogenetic stimuli, but they have the same average core temperature as young people.
Body temperature is closely regulated through a balance between heat production and heat dissipation.2 The majority of endogenous heat production results from metabolic activity in the heart and liver.(????) The skin accounts for 90% of heat loss and the lungs contribute the rest.
Radiation cooling (heat loss in the form of infrared radiation) is the primary method of thermal load dissipation and accounts for approximately 60% of heat loss. Conduction (direct transfer of heat to a cooler object) and convection (heat removal by air currents) account for 10% to 15% of heat loss while evaporation from skin and the respiratory tract accounts for 25% to 30% of heat loss. Conduction is an important mechanism of heat loss in immersion accidents because thermal conduction of water is approximately 30 times that of air. Convection is important in windy conditions by removing the warm insulating layer of air around the body. The preoptic nucleus of the anterior hypothalamus is the thermal control center, which maintains body temperature at a given set value. In response to a decrease in core body temperature, the hypothalamus initiates mechanisms to conserve heat by cutaneous vasoconstriction and produce heat by stimulation of muscular activity in the form of shivering.2,3 Nonshivering thermogenesis occurs via increased activity of thyroxine and catecholamines.4,5 In a conscious individual, the appreciation of cold induces the individual to exercise, wear more clothes, or move to a warmer environment.
As the core temperature decreases below 35°C (95°F), the coordinated systems responsible for thermoregulation begin to fail because the physiologic responses to minimize heat loss are very limited.6 Primary hypothermia (accidental hypothermia) refers to a spontaneous decrease of core body temperature, usually as result of exposure to cold environments without adequate protection. Environmental hypothermia results from a combination of heat loss by convection (degree of wind exposure), conduction, and radiation to the surrounding ambient air.Secondary hypothermia represents a complication of an underlying disorder. Some of the disorders and conditions that may predispose an individual to hypothermia by decreasing heat production, increasing heat loss, or interfering with the central or peripheral control of thermoregulation are listed in Table 131-1.1-3,6-10
Humans are capable of significant heat generation. Strenuous exercise can increase heat generation twentyfold.
Normally, the body dissipates heat into the environment via four mechanisms. The evaporation of skin moisture is the single most efficient mechanism of heat loss but becomes progressively ineffective as the relative humidity rises to >70%. The radiation of infrared electromagnetic energy directly into the surrounding environment occurs continuously. (Conversely, radiation is a major source of heat gain in hot climates.) Conduction— the direct transfer of heat to a cooler object—and convection—the loss of heat to air currents—become ineffective when the environmental temperature exceeds the skin temperature.
Factors that interfere with the evaporation of diaphoresis significantly increase the risk of heat illness. Examples include dripping of sweat off the skin, constrictive or occlusive clothing, dehydration, and excessive humidity. While air is an effective insulator, the thermal conductivity of water is 25 times greater than that of air at the same temperature. The wet-bulb globe temperature is a commonly used index to assess the environmental heat load. This calculation considers the ambient air temperature, the relative humidity, and the degree of radiant heat.
The regulation of this heat load is complex and involves the central nervous system (CNS), thermosensors, and thermoregulatory effectors. The central thermostat activates the effectors that produce peripheral vasodilation and sweating. The skin surface is in effect the radiator and the principal location of heat loss, since skin blood flow can increase 25–30 times over the basal rate. This dramatic increase in skin blood flow, coupled with the maintenance of peripheral vasodilation, efficiently radiates heat. At the same time, there is a compensatory vasoconstriction of the splanchnic and renal beds.
Acclimatization to heat reflects a constellation of physiologic adaptations that permit the body to lose heat more efficiently. This process often requires one to several weeks of exposure and work in a hot environment. During acclimatization, the thermoregulatory set point is altered, and this alteration affects the onset, volume, and content of diaphoresis. The threshold for the initiation of sweating is lowered, and the amount of sweat increases, with a lowered salt concentration. Sweating rates can be 1–2 L/h in acclimated individuals during heat stress. Plasma volume expansion also occurs and improves cutaneous vascular flow. The heart rate lowers, with a higher stroke volume. After the individual leaves the hot environment, improved tolerance to heat stress dissipates rapidly, the plasma volume decreases, and de-acclimatization occurs within weeks.
A normal body temperature is ordinarily maintained despite environmental variations because the hypothalamic thermoregulatory center balances the excess heat production derived from metabolic activity in muscle and the liver with heat dissipation from the skin and lungs. According to studies of healthy individuals 18–40 years of age, the mean oral temperature is 36.8° ± 0.4°C (98.2° ± 0.7°F), with low levels at 6 a.m. and higher levels at 4–6 p.m. The maximal normal oral temperature is 37.2°C (98.9°F) at 6 a.m. and 37.7°C (99.9°F) at 4 p.m.; these values define the 99th percentile for healthy individuals. In light of these studies, an a.m. temperature of >37.2°C (>98.9°F) or a p.m. temperature of >37.7°C (>99.9°F) would define a fever. The normal daily temperature variation is typically 0.5°C (0.9°F). However, in some individuals recovering from a febrile illness, this daily variation can be as great as 1.0°C. During a febrile illness, the diurnal variation is usually maintained, but at higher, febrile levels. The daily temperature variation appears to be fixed in early childhood; in contrast, elderly individuals can exhibit a reduced ability to develop fever, with only a modest fever even in severe infections.
Rectal temperatures are generally 0.4°C (0.7°F) higher than oral readings. The lower oral readings are probably attributable to mouth breathing, which is a factor in patients with respiratory infections and rapid breathing. Lower-esophageal temperatures closely reflect core temperature. Tympanic membrane thermometers measure radiant heat from the tympanic membrane and nearby ear canal and display that absolute value (unadjusted mode) or a value automatically calculated from the absolute reading on the basis of nomograms relating the radiant temperature measured to actual core temperatures obtained in clinical studies (adjusted mode). These measurements, although convenient, may be more variable than directly determined oral or rectal values. Studies in adults show that readings are lower with unadjusted-mode than with adjusted-mode tympanic membrane thermometers and that unadjusted-mode tympanic membrane values are 0.8°C (1.6°F) lower than rectal temperatures.
In women who menstruate, the a.m. temperature is generally lower in the 2 weeks before ovulation; it then rises by ∼0.6°C (1°F) with ovulation and remains at that level until menses occur. Body temperature can be elevated in the postprandial state. Pregnancy and endocrinologic dysfunction also affect body temperature.
Content 6
{The hypothalamus integrates information from the environment, nervous system, and the other organ systems.}
3) Corticotrophin Releasing Hormone is released by the paraventricular nucleus and taken up by the portal system where it has its action on the anterior lobe of the pituitary. A recent, interesting development is the finding of a direct projection from the eye to the suprachiasmatic nucleus of the supraoptic hypothalamic region.
4) The median eminence—the bulge from which the infundibulum extends to the anterior pituitary . In the median eminence, a dense network of capillaries arises from the superior hypophyseal arteries. These capillaries drain into portal vessels that traverse the pituitary stalk and then form a capillary network within the anterior pituitary gland. The primary direction of this hypophyseal portal system is from hypothalamus to pituitary, however, retrograde flow also exists. This creates an ultrashort feedback loop between the pituitary gland and hypothalamic neurons.
5) The mammillary or posterior region at the level of the mammillary bodies [What does it secrete?]
6). Thermoregulation
sexually dimorphic nucleus (SDN, intermediate nucleus, INAH-1),
suprachiasmatic nucleus (SCN)
tuberal lateral nucleus (NTL).
The hypothalamus regulates body functions that vary at different times of the day (e.g., body temperature, hormone secretion, hunger) or those that vary over a period of many days (e.g., menstrual cycle). A projection from the retina to the suprachiasmatic nucleus is thought to supply the clock with day-night information needed for synchronizing diurnal (daily) rhythms (also known as circadian rhythms). Lesions of the hypothalamus often disrupt the state of the sleep-waking cycle.
This neural region is associated with hormonally related behaviour (anger and sexual activity), homeostatic regulation (blood pressure, heart rate, appetite, and temperature) and functions that relate to both (puberty, reproductive cycles, and lactation). The hypothalamus is a small part of the diencephalon that is involved
in the mediation of endocrine, autonomic and behavioral functions.book whenever the specific hormone systems to which they belong are described. Several neuropeptides have been isolated from the hypothalamus, and many continue to be discovered. However, only those that have been demonstrated to control anterior pituitary function (hypophysiotropic hormones) and, therefore, play an important role in endocrine physiology will be discussed.
GnRH is a small peptide produced in the hypothalamus by specialized nerve cells; as such, GnRH is called a neurohormone, a class of hormones that include thyrotropin-releasing hormone, oxytocin (see below), antidiuretic hormone (Chapter 18), and corticotropin-releasing hormone. Release of GnRH results in activation of a specific GnRH receptor, located in the gonadotropes of the pituitary gland. This receptor is a membrane-bound G-protein-coupled stimulator of phospholipase C, which results in calcium release and protein kinase C activation via conversion of plasma membrane phosphatidylinositol into inositol triphosphate and diacylglycerol (Chapter 8). These signals result in production and release of FSH and LH, as will be described below. Regulation of this important signal is multifold. GnRH is degraded within minutes so it is constantly produced in pulses, and the size and frequency of these pulses is important in signaling. These GnRH pulses are constant in males but vary in females, depending on the menstrual cycle. Interestingly, the frequency of the GnRH pulses result in different expression of FSH (low frequency) and LH (high frequency). As a result FSH and LH are variably expressed during the female menstrual period (Figure 20-2). Levels of testosterone, estrogen, and prolactin (increased during pregnancy) as well as increased concentration of FSH and LH create a negative feedback loop, which can decrease GnRH pulses.
(2) involved in temperature regulation, homeostatic regulation (blood pressure, heart rate, appetite, and temperature
(3) control of food and water intake (thirst),
4) sexual behavior and reproduction
(5) control of daily cycles in physiological state and behavior
(6) mediation of emotional responses,such as anger
(7) controls functions that relate to both (puberty, reproductive cycles, and lactation).
03. Hypothalamic–Pituitary–Adrenal Axis
04. Hypothalamic–Pituitary–Gonadal Axis
http://nba.uth.tmc.edu/neuroscience/s4/chapter01.html
- Chapter 1: Hypothalamus: Structural Organization, Patrick Dougherty, Ph.D.
- Chapter 2: Hypothalamic Control of Pituitary Hormones, Patrick Dougherty, Ph.D.
- Chapter 3: Central Control of the Autonomic Nervous System & Thermoregulation, Patrick Dougherty, Ph.D.
- Chapter 4: Central Control of Feeding Behavior, Patrick Dougherty, Ph.D.
- Chapter 5: Limbic System: Hippocampus, Anthony Wright, Ph.D.
- Chapter 6: Limbic System: Amygdala, Anthony Wright, Ph.D.
- Chapter 7: Learning and Memory, John H. Byrne, Ph.D.
- Chapter 8: Higher Cortical Functions: Language, Anthony Wright, Ph.D.
- Chapter 9: Higher Cortical Functions: Association and Executive Processing, Anthony Wright, Ph.D.
- Chapter 10: CNS Aging and Alzheimer's Disease, Jack Waymire, Ph.D.
- Chapter 11: Blood Brain Barrier and Cerebral Metabolism, Pramod Dash, Ph.D.
- Chapter 12: Neurotransmitter and Cell Death, Pramod Dash, Ph.D.
-
Oxytocin, and Mother-Baby Interactions
POSTED ON TUE, OCT 29, 2019 @ 01:00 PM BY LAURA SNIDER
The fact that it’s good for parents to spend quality time with their children is just common sense—but did you know that a new study showed that mothers’ behavior has an effect on the development of their babies’ oxytocin systems? A team of scientists from the University of Virginia and the Max Planck Institute for Human Cognitive and Brain Sciences found that “greater maternal involvement seems to have the potential to upregulate the oxytocin system in human offspring.” Before we go into exactly what that means, though, let’s talk about oxytocin for a second.
You may have heard of oxytocin before: people sometimes call it the “love hormone” or the “cuddle hormone.” It’s a versatile hormone, released in response to the activation of sensory nerves. During childbirth, it causes smooth muscle in the uterus to contract, and during breastfeeding, it stimulates milk ejection from the mammary glands. It’s released during sexual intercourse, skin-to-skin contact between mothers and infants, and positive human-human or human-animal social interactions (interactions between humans and dogs, for instance).
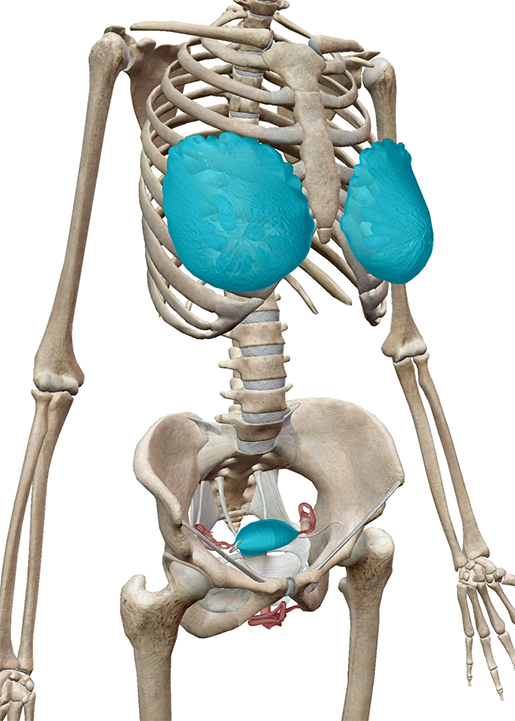 The mammary glands (breast tissue) and uterus of the female reproductive system.
The mammary glands (breast tissue) and uterus of the female reproductive system.
Image from Human Anatomy Atlas.Want to know something else cool about oxytocin? It functions as both a hormone and a neurotransmitter.
Oxytocin is produced by the hypothalamus, and the posterior pituitary gland sends it off into systemic circulation to perform its childbirth and labor functions. Once oxytocin is out of the brain, it can’t get back in (due to the blood-brain barrier).
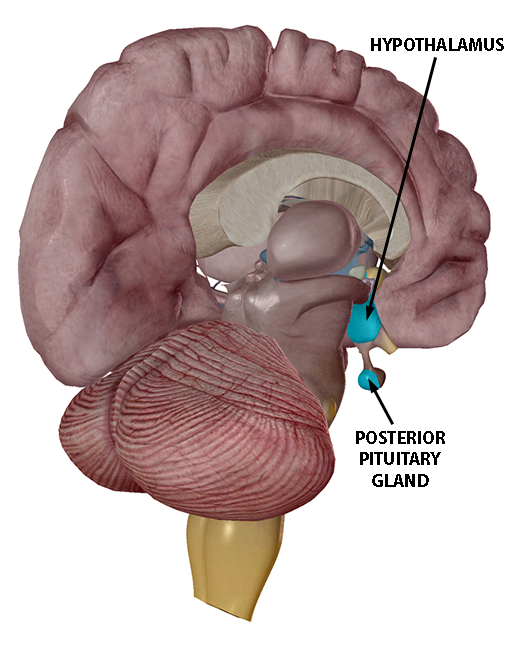 Image from Human Anatomy Atlas.
Image from Human Anatomy Atlas. However, oxytocin acts within the brain as well, affecting the CNS. Oxytocinergic neurons (neurons that produce oxytocin as a neurotransmitter) project from the paraventricular nucleus (PVN) to important regulatory brain areas, which include “other nuclei within the hypothalamus, the amygdala, the hippocampus, the PAG, the frontal cortex, the raphe nuclei, the striatum, the [nucleus accumbens], the vagal nuclei (both the nucleus of the solitary tract (NTS) and the dorsal vagal nucleus (DMX), and the [locus coeruleus].”
The UVA/Max Planck study investigated whether babies had lower methylation of the oxytocin receptor gene (the gene that codes for a protein that serves as a receptor for oxytocin) when their mothers were more engaged during play. DNA methylation is an epigenetic process, meaning that it changes the expression of genes but doesn’t alter their sequence of nucleotides. DNA methylation usually represses gene expression.
Here’s how the study worked. Mothers and their five-month-old babies were recorded during a play session and rated on both maternal and infant factors. Maternal factors included how talkative the mother was, how close she was to the baby, how attentive the mother was to the baby’s needs, how positive the mother’s mood was, and the duration of passive and active touch. Infant factors included how attentive the baby was, how positive their mood was, and the duration of the baby’s smiles with and without vocalizations.
The mothers also filled out a breastfeeding questionnaire, a maternal demographics survey, a short form of the social support questionnaire (SSQ6), the Parenting Sense of Competence Scale, and the Edinburgh Postnatal Depression Scale. Keep in mind: this was an observation, not an experimental manipulation.
Saliva samples were collected when the children were five months old (this was when the play sessions were recorded as well), and then around a year later, when the children were 18 months old.
Ultimately, the study found that there was less methylation of the oxytocin receptor gene at 18 months in mother-baby pairs where the mother was more engaged during play at 5 months. The mothers’ levels of OXTR methylation did not change significantly between the 5- and 18-month samples. On the basis of these results, the authors suggest that “infancy may provide a sensitive period in development during which the oxytocin system is dynamic, plastic, and sensitive to the social environment.”
Reference
https://human-memory.net/hypothalamus/
- Identify the two hormones released from the posterior pituitary, their target cells, and their principal actions
- Identify the six hormones produced by the anterior lobe of the pituitary gland, their target cells, their principal actions, and their regulation by the hypothalamus
- ++++++++++++++++++++++++++++++
-
Nervous connections
The nervous connections can be divided into afferent and efferent fibers.
Afferent fibers

Hypothalamus receives afferent fibers carrying somatic and visceral sensations as well as from special senses. Following are the important afferents of hypothalamus:
- Somatic and visceral afferents via lemniscal afferent fibers and nucleus of tractus solitarius, that reach the hypothalamus via reticular formation
- Visual afferents from the optic chiasma reach the suprachiasmatic nucleus
- Olfactory afferents are received through medial forebrain bundle
- Auditory afferents though not identified completely but are influenced by the hypothalamus
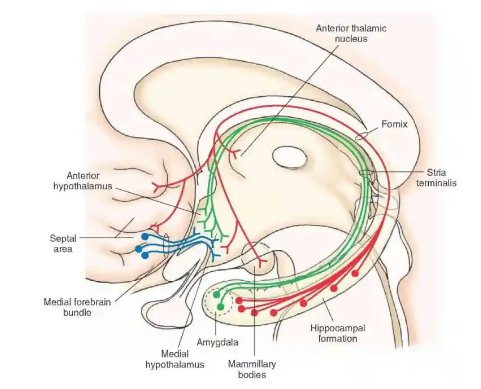
- Hippocampo-hypothalamic afferents reach via fornix to mamillary bodies
- Tegmental fibers from midbrain
- Thalamo-hypothalamic fibers from the midline and dorsomedial nuclei of the thalamus
- Amygdalo-hypothalamic fibers from the amygdaloid complex reach the hypothalamus via stria terminalis
Efferent fibers

The efferent connections of hypothalamic nuclei are also complex and numerous. Here, we will mention some important connections.
- To brain stem and spinal cord: The hypothalamic nuclei send efferent fibers to nuclei present in the brainstem and spinal cord. In this way, they control the autonomic nervous system.
- Mammillothalamic Tract: This tract consists of fibers arising in the mamillary body and terminating in the anterior nucleus of thalamus.
- Mammillotegmental Tract: These fibers terminate in the reticular formation, present in the tegmentum of the midbrain.
- Limbic System: The nuclei in the hypothalamus also send efferent fibers to the various nuclei of the limbic system.
Hypothalamus also uses nervous connections to communicate with the pituitary gland. The cell bodies of these neurons are present in the hypothalamus and the axonal terminals in the posterior pituitary gland.
These neurons synthesize the oxytocin and vasopressin hormones, which are stored in the axonal terminals in the posterior pituitary and are released on demand.
Autonomic Control
The most important function of the hypothalamus is to integrate the endocrine system and the autonomic nervous system. Hypothalamus acts as a higher center for controlling the autonomic functions of the brain stem and spinal cord.
The stimulation of the posterior and lateral nuclei of the hypothalamus has been shown to cause a sympathetic response. On the other hand, the stimulation of the anterior nucleus and the preoptic area influences parasympathetic responses in the body.
Endocrine Control

Hypothalamus produces releasing factors or inhibitory factors for controlling the hormones released by the pituitary gland. These factors include:
- Growth hormone-releasing hormone and inhibiting hormone also called somatostatin
- Prolactin releasing hormone and inhibiting hormone
- Corticotropin-releasing hormone
- Thyrotropin-releasing hormone
- Luteinizing hormone-releasing hormone
These factors promote or inhibit the release of hormones from the anterior pituitary. The release of these factors from the hypothalamus is controlled by positive and negative feedback mechanisms depending on the levels of a particular hormone in blood.
Secretion of Hormones

Hypothalamus not only secretes the regulating factors but also secretes two important hormones; vasopressin and oxytocin. Although these hormones are released from the posterior pituitary, they are actually produced by the neurons in the hypothalamus and are stored in the axonal endings present in the posterior pituitary.
Temperature Regulation
Temperature regulation is another important function of the hypothalamus.
The anterior part of the hypothalamus controls processes that dissipate heat from the body. Its stimulation causes dilation of blood vessels and sweating, which causes a decrease in body temperature.
Contrary to this, stimulation of the posterior part of the hypothalamus results in vasoconstriction of the skin blood vessels and inhibition of sweating resulting in conservation of body temperature.
Regulation of Food and Water Intake
Hypothalamus is also the site of the hunger center and satiety center in the brain.
The stimulation of the lateral region of the hypothalamus stimulates hunger and results in the intake of food. This region is termed as hunger center.
Controlling Emotions and Behavior
Being a part of the limbic system hypothalamus also controls the emotions and behavior of a person. It is believed that the hypothalamus integrates all the afferent information from other areas of the brain and brings about the physical expression of emotion.
Stimulation of the lateral area of the hypothalamus is associated with the feelings of rage whereas the stimulation of the medial area results in feelings of passivity.
Clinical Significance
As stated earlier, the hypothalamus controls almost all the processes in the body. The lesions of the hypothalamus may be due to inflammation, tumor or vascular disorder. It may also be compressed by the tumors of the surrounding region. Any lesion of the hypothalamus can result in the following clinical disorders.
Obesity and Wasting
Severe obesity can occur in case of hypothalamic lesions associated with genital hypoplasia and atrophy.
Sexual disorders
In children, hypothalamic lesions can cause sexual retardation. After puberty, they can result in impotence or amenorrhea.
Hyperthermia and Hypothermia
Depending on the part of hypothalamus damaged by a lesion, hypothalamic lesions can result in hypothermia or hyperthermia.
Diabetes Insipidus
It results from the lesion of the suprachiasmatic nucleus present in the hypothalamus. It may also be due to the destruction of nervous pathways from the hypothalamus to the posterior pituitary.
Emotional disturbance
Hypothalamic lesions can also cause emotional disturbances that can result in unexplained attacks of weeping, laughter, uncontrollable rage, excessive maniac outbursts and depressive reactions etc.
Conclusion/Summary
Hypothalamus is an important part of forebrain located just below the thalamus.
It consists of groups of neurons that are divided into two-zone, lateral zone and medial zone.
Both zones have their own nuclei. However, nuclei like the suprachiasmatic nucleus and preoptic nucleus overlap both the zones.
Hypothalamus receives afferent fibers from almost all regions of the body and brain. It receives somatic and visceral sensory afferents, visual and auditory afferents, afferent fibers related to smell, emotions and feelings.
It sends efferent fibers to the brainstem and spinal cord, thalamus, tegmentum, and the limbic system. It is also considered to be a part of the limbic system.
Hypothalamus communicates to the rest of the body via nervous and endocrine connections and cerebrospinal fluid.
The important functions performed by hypothalamus include:
- Control of autonomic nervous system
- Control of hormones released by the anterior pituitary
- Secretion and synthesis of Vasopressin and Oxytocin
- Control of body temperature
- Regulation of food and water intake
Hypothalamic lesions can result in following clinical disorders:
- Weight loss and wasting
- Sexual disorders
- Emotional disorders
- Diabetes insipidus
- Hypothermia or hyperthermia
References
- Dr. Boeree, C. George. “The Emotional Nervous System”. The Limbic System. Retrieved 18 April 2016.
- “NCI Dictionary of Cancer Terms”. National Cancer Institute.
- Melmed, S; Polonsky, KS; Larsen, PR; Kronenberg, HM (2011). Williams Textbook of Endocrinology (12th ed.). Saunders. p. 107. ISBN 978-1437703245
The hypothalamus is a small deeply located region placed at the crossroad of neurovegetative, neuroendocrine, limbic, and optic systems.
Although deep brain stimulation techniques have proven that it could be feasible to modulate these systems, targeting the hypothalamus and in particular specific nuclei and white bundles, is still challenging. Our goal was to make a synthesis of relevant topographical data of the human hypothalamus, under the form of magnetic resonance imaging maps useful for mastering its elaborated structure as well as its neighborhood. As from 1.5 Tesla, Inversion-Recovery sequence allows locating the hypothalamus and most of its components. Spotting hypothalamic compartments is possible according to specific landmarks: the anterior commissure, the mammillary bodies, the preoptic recess, the infundibular recess, the crest between the preoptic and the infundibular recesses, the optical tract, the fornix, and the mammillo-thalamic bundle. The identification of hypothalamus and most of its components could be useful to allow the quantification of local pathological processes and to target specific circuitry to alleviate severe symptoms, using physical or biological agents.
Keywords: Brain mapping, hypothalamus, inversion-recovery sequence, magnetic resonance imaging, stereotaxyStructure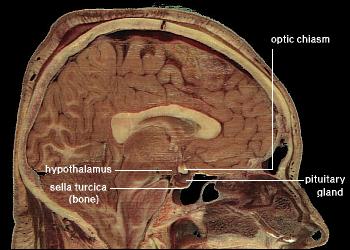
The hypothalamus is a small and heterogeneous region of the prosencephalon.

[Its numerous functional facets, from behavior to chronobiology, make the hypothalamus likely one of the most challenging region of the human brain. From a clinical perspective, several reports have proven that hypothalamus neuromodulation is possible;[10,12,23,26,33,34,50] however, the most important part of the clinical research has to be done before proposing reliable long-term invasive functional treatments.
Paradoxically although the hypothalamus was extensively explored in several species, it is still a poorly known region of the deep human brain. Pioneer's histological atlas provided details that are often difficult to relate to the clinical context. Our goal was to make a synthesis of relevant topographical data of the human hypothalamus, under the form of maps useful for mastering its elaborated structure. Magnetic resonance imaging (MRI) was used to create these maps, since it is the most advanced way to finely explore in a noninvasively the hypothalamus. This was accomplished using the widely available 1.5 Tesla device. Anatomical landmarks were defined to map structures to subject's peculiarities, providing means to further research protocols.
Function
++++++++++++++++++++
https://nba.uth.tmc.edu/neuroscience/s4/chapter01.html
++++++++++++++
The hypothalamus regulates of homeostasis essential for survival and reproduction of the species.
The importance of this function is underscored by the structural organization and connectivity of the hypothalamus as almost every major subdivision of the neuraxis communicates with the hypothalamus and is subject to its influence.
Reference: https://nba.uth.tmc.edu/neuroscience/toc.htm
- Secrete ADH (Vasopressin)
- Secrete Oxytocin
- Growth Hormone-Releasing Hormone
- Stimulate Thirst
- Hypothalamus-Pituitary-Thyroid Axis
- Temperature Regulation
- Secrete Gonadotrophin
Vasopressin (AVP) is synthesized in magnocellular neurons (Supraoptic nuclei); the distal axons of these neurons project to the posterior pituitary, from which AVP is released into the circulation. Argine vasopressin and oxytocin are stored in Herring bodies at the axon terminals in the posterior pituitary where they are released into the blood stream. These neurons are sensitive to changes in extracellular osmolality. When ECF osmolality increases, these cells shrink and release ADH from the posterior pituitary.
A network of central “osmoreceptor” neurons, which includes the AVP-expressing magnocellular neurons themselves, sense circulating osmolality via nonselective, stretch-activated cation channels.
These osmoreceptor neurons are activated or inhibited by modest increases and decreases in circulating osmolality, respectively; activation leads to AVP release and thirst.
AVP secretion is stimulated as systemic osmolality increases above a threshold level of ~285 mOsm/kg, above which there is a linear relationship between osmolality and circulating AVP.
Thirst and thus water ingestion are also activated at ~285 mOsm/kg, beyond which there is an equivalent linear increase in the perceived intensity of thirst as a function of circulating osmolality.
Effects of ADH
1. On kidney - ADH markedly increases water reabsorption in medullary collecting tubules, which tends to reduce plasma osmolality back to normal.
Conversely, a decrease in extracellular osmolality causes osmoreceptors to swell and suppresses the release of ADH. Decreased ADH secretion allows a water diuresis, which tends to increase osmolality to normal. Peak diuresis occurs once circulating ADH is metabolized (90-120 min). With complete suppression of ADH secretion, the kidneys can excrete up to 10-20 L of water per day.
[Damage to the anterior hypothalamus blocks the production of ADH, resulting in diabetes insipidus, which is characterized by rapid water loss from the kidneys.1]
[Nonosmotic Release of Antidiuretic Hormone
The carotid baroreceptors and probably atrial stretch receptors can also stimulate ADH release following a 5-10% decrease in blood volume. Other nonosmotic stimuli include pain, emotional stress, and hypoxia.]
Oxytocin hormone produce in the Paraventricular nucleus ,at the end of pregnancy, is produced in large amount and stimulates labor contraction of the uterus
These axons (likely, but dendrites have not been ruled out) have collaterals that innervate oxytocin receptors in the nucleus accumbens.
The peripheral hormonal and behavioral brain effects of oxytocin are thought to be coordinated through its common release through these collaterals.[12]Oxytocin is also made by some neurons in the paraventricular nucleus that project to other parts of the brain and to the spinal cord.[111] Depending on the species, oxytocin receptor-expressing cells are located in other areas, including the amygdala and bed nucleus of the stria terminalis.
In the pituitary gland, oxytocin is packaged in large, dense-core vesicles, where it is bound to neurophysin I as shown in the inset of the figure; neurophysin is a large peptide fragment of the larger precursor protein molecule from which oxytocin is derived byenzymatic cleavage.
Secretion of oxytocin from the neurosecretory nerve endings is regulated by the electrical activity of the oxytocin cells in the hypothalamus. These cells generate action potentials that propagate down axons to the nerve endings in the pituitary; the endings contain large numbers of oxytocin-containing vesicles, which are released by exocytosis when the nerve terminals are depolarised.
growth hormone-releasing hormone
Osmoreceptors in the lateral preoptic area of the hypothalamus are very sensitive to changes in extracellular osmolality. Activation of these neurons by increases in ECF osmolality induces thirst and causes the individual to drink water.
Conversely, hypoosmolality suppresses thirst.
Thirst is the major defense mechanism against hyperosmolality and hypernatremia, because it is the only mechanism that increases water intake.
The hypothalamic thermoregulatory center balances excess heat production from metabolic activity in muscle and liver with heat dissipation from the skin and lungs to maintain a normal body temperature of 36.8° ± 0.4°C (98.2° ± 0.7°F), with diurnal variation (lower in a.m., higher in p.m.).
The hypothalamus maintains core body temperature within very narrow range.
- At one end is the threshold for sweating and vasodilation with heat dissipation from the skin and lungs..
- At the other is the threshold for vasoconstriction and shivering (metabolic activity in muscle and the liver)
The human body generates enough heat, even at rest, to raise the body temperature by 1°C every hour.
A normal body temperature is maintained despite environmental variations because the hypothalamic thermoregulatory center balances the excess heat production derived from metabolic activity in muscle (shivering) and the liver
The system is effective such that the daily variation in body temperature is only ±0.6°C (2).
Neurons in both the preoptic anterior hypothalamus and the posterior hypothalamus receive two kinds of signals:
- one from peripheral nerves that transmit information from warmth/cold receptors in the skin
- the other from the temperature of the blood bathing the region.
These two types of signals are integrated by the thermoregulatory center of the hypothalamus to maintain normal temperature.
Increasing core temperature a fraction of a degree induces sweating and vasodilation, whereas a minimally reduced core temperature triggers vasoconstriction and shivering.1
++++++++++++++++++++++++++++++++++++++About 90% of the excess heat generated by metabolism is dissipated through the skin, with the remainder being lost in exhaled gas.
Heat exchange between the body and its surroundings is accomplished in several ways:
RadiationRadiation refers to the loss of heat via infrared heat rays (a type of electromagnetic wave) that radiate out from the skin. These waves emanate from all objects that exist above absolute zero temperature, and the intensity of radiation increases as the temperature of the object increases.-----------------------------------------------------------------------------------------
Under normal conditions, radiation accounts for about 60% of the heat loss from the human body (3).ConductionConduction is the transfer of heat from an object of higher temperature to an object of lower temperature. This is the behavior of heat as kinetic energy, which imparts motion to molecules and results in the transfer of heat from a hotter to a colder object. Heat transfer by conduction alone is responsible for only about 15% of heat loss from the body (2).ConvectionWhen heat is lost from the skin, it warms the air just above the skin surface. This increase in surface temperature limits the further loss of body heat by conduction. However, when an air current from a fan (or a gust of wind) is passed across the skin, it displaces the warm layer of air above the skin and replaces it with cooler air, and this process facilitates the continued loss of body heat by conduction. The same effect is produced by increases in blood flow just underneath the skin. The action of currents (air and blood) to promote heat loss is known as convection.EvaporationThe transformation of water from liquid to gaseous phase requires heat (called the latent heat of vaporization), and when water or sweat evaporates from the surface of the body, the heat that is utilized is body heat. Normally, evaporation accounts for about 20% of the loss of body heat (mostly as a result of insensible fluid losses from the lungs). Evaporation plays a much greater role in the adaptation to thermal stress (see next).The maintenance of body temperature in conditions of thermal stress (hot weather, strenuous exercise, or both) is primarily achieved by enhanced blood flow to the skin (convective heat loss) and the loss of sweat (evaporative heat loss).The Role of SweatingThe evaporation of sweat from the skin is responsible for at least 70% of the loss of body heat during periods of thermal stress. The evaporation of one liter of sweat from the skin is accompanied by the loss of 580 kilocalories (kcal) of heat from the body (3). This is about one-quarter of the daily heat production by an average-sized adult at rest. Thermal sweating (as opposed to “nervous sweating”) can achieve rates of 1 to 2 liters per hour (3), which means that over 1,000 kcal of heat can be lost hourly during profuse sweating. It is important to emphasize that sweat must evaporate to ensure loss of body heat. Wiping sweat off the skin will not result in heat loss, so this practice should be discouraged during strenuous exercise.P.699According to studies of healthy individuals 18–40 years of age, the mean oral temperature is 36.8° ± 0.4°C (98.2° ± 0.7°F), with low levels at 6 a.m. and higher levels at 4–6 p.m.
The maximal normal oral temperature is 37.2°C (98.9°F) at 6 a.m. and 37.7°C (99.9°F) at 4 p.m.; these values define the 99th percentile for healthy individuals. In light of these studies, an a.m. temperature of >37.2°C (>98.9°F) or a p.m. temperature of >37.7°C (>99.9°F) would define a fever. The normal daily temperature variation is typically 0.5°C (0.9°F). However, in some individuals recovering from a febrile illness, this daily variation can be as great as 1.0°C. During a febrile illness, the diurnal variation is usually maintained, but at higher, febrile levels. The daily temperature variation appears to be fixed in early childhood; in contrast, elderly individuals can exhibit a reduced ability to develop fever, with only a modest fever even in severe infections.
Rectal temperatures are generally 0.4°C (0.7°F) higher than oral readings. The lower oral readings are probably attributable to mouth breathing, which is a factor in patients with respiratory infections and rapid breathing. Lower-esophageal temperatures closely reflect core temperature. Tympanic membrane thermometers measure radiant heat from the tympanic membrane and nearby ear canal and display that absolute value (unadjusted mode) or a value automatically calculated from the absolute reading on the basis of nomograms relating the radiant temperature measured to actual core temperatures obtained in clinical studies (adjusted mode). These measurements, although convenient, may be more variable than directly determined oral or rectal values. Studies in adults show that readings are lower with unadjusted-mode than with adjusted-mode tympanic membrane thermometers and that unadjusted-mode tympanic membrane values are 0.8°C (1.6°F) lower than rectal temperatures.
In women who menstruate, the a.m. temperature is generally lower in the 2 weeks before ovulation; it then rises by ∼0.6°C (1°F) with ovulation and remains at that level until menses occur. Body temperature can be elevated in the postprandial state. Pregnancy and endocrinologic dysfunction also affect body temperature.
In a neutral temperature environment, the human metabolic rate produces more heat than is necessary to maintain the core body temperature in the range of 36.5–37.5°C (97.7–99.5°F).
These cytokines stimulate the immune responses of T and B cells, macrophages, and polymorphonuclear leukocytes. They appear to act via a common mechanism involving the induction of prostaglandin synthesis. Feedback inhibitory responses are mediated by adrenocorticotropic hormone, arginine vasopressin, serotonin, dopamine, and other homeostatic mechanisms, thus emphasizing the orchestrated nature of fever production and response to infection. These thermoregulatory mechanisms rarely allow fevers to exceed 41° C (106° F). Temperatures exceeding 41° C are often due to a drug-induced imbalance in these mechanisms and may cause direct cellular damage.
The hypothalamus contains temperature-sensitive neurons that have receptors for pro-inflammatory and anti-inflammatory cytokines, which are continuously balanced to maintain a homeothermic set point. When body temperature becomes elevated, cutaneous vasodilation and sweating occur, and people may reduce activity and seek a cooler environment. In contrast, low body temperature is increased by shivering, piloerection, cutaneous vasoconstriction, adding clothes, and seeking a warmer environment. In a febrile illness, symptoms may be due to the underlying disease or to the fever itself. Malaise is the rule, and many febrile patients experience myalgia secondary to the muscle contractions used to generate temperature elevation. Although it was once thought that the back and thigh pain related to rigors suggests bacteremia, any febrile stimulus can produce such symptoms. The chill associated with rigors may be related to the surface vasoconstriction that accompanies the increase in core temperature.
Bacteria release cell wall products such as peptidoglycans and endotoxin, as well as enterotoxins, which bind to Toll-like receptors (TLRs) on phagocytes (neutrophils, macrophages) and endothelial cells. As a result, pyrogenic cytokines such as interleukin (IL)-1 and IL-6 or tumor necrosis factor (TNF)-α are released into the circulation and bind to cytokine receptors in the hypothalamic organum vasculosum of lamina terminalis (OVLT). Bacterial products may also directly bind to TLRs on the OVLT. Activation of TLRs and cytokine receptors induces cyclooxygenase 2 (COX2), which leads to the production of prostaglandin E2 (PGE2) and elevates cyclic adenosine monophosphate (cAMP) in the brain. This, in turn, triggers neurons in the thermoregulatory center to raise the hypothalamic thermostatic set point. In addition, neuronal signals to the cortex prompt behavioral changes to conserve heat (e.g., posturing, adding clothing). The hypothalamus also triggers sympathetic peripheral efferent nerves that constrict peripheral blood vessels and conserve central heat until hypothalamic PGE2 levels fall.
Among the broad range of conditions that cause fever are infections, malignancies, autoimmune diseases, metabolic diseases, chronic inflammatory conditions, medications (including immunizations), CNS abnormalities, and exposure to excessive environmental heat. (How do these produced pyrogens??)
The hypothalamic set point increases, causing peripheral vasoconstriction (i.e., heat conservation). [The patient feels cold as a result of blood shunting to the internal organs.ref?] Mechanisms of heat production (e.g., shivering, increased hepatic thermogenesis) help to raise the body temperature to the new set point. Increases in peripheral prostaglandin E2 account for the nonspecific myalgias and arthralgias that often accompany fever. When the set point is lowered again by resolution or treatment of fever, processes of heat loss (e.g., peripheral vasodilation and sweating) commence.
Anesthetic agents inhibit central thermoregulation by interfering with these hypothalamic reflex responses. For example,isoflurane produces a dose-dependent decrease in the threshold temperature that triggers vasoconstriction (3°C decrease for each percent of inhaled isoflurane). Both general and regional anesthetics increase the interthreshold range, albeit by different mechanisms. Spinal and epidural anesthetics, like general anesthetics, lead to hypothermia by causing vasodilation and internal redistribution of heat. The accompanying thermoregulatory impairment from regional anesthetics that allows continued heat loss is likely due to altered perception by the hypothalamus of temperature in the anesthetized dermatomes rather than a central drug effect, as with general anesthetics.
Moderate activity increases thermogenesis and results in a transiently increased temperature until heat-dissipating processes are engaged. Each 1° F increase in temperature results in a 7% increase in the basal metabolic rate. Vaporization from the lungs and skin accounts for a third of basal body heat loss and for as much as all heat loss at ambient dry temperatures above 36° C. The elderly have a decrease in basal metabolism, as well as blunted responses to thermogenetic stimuli, but they have the same average core temperature as young people.
Body temperature is closely regulated through a balance between heat production and heat dissipation.2 The majority of endogenous heat production results from metabolic activity in the heart and liver.(????) The skin accounts for 90% of heat loss and the lungs contribute the rest.
Radiation cooling (heat loss in the form of infrared radiation) is the primary method of thermal load dissipation and accounts for approximately 60% of heat loss. Conduction (direct transfer of heat to a cooler object) and convection (heat removal by air currents) account for 10% to 15% of heat loss while evaporation from skin and the respiratory tract accounts for 25% to 30% of heat loss. Conduction is an important mechanism of heat loss in immersion accidents because thermal conduction of water is approximately 30 times that of air. Convection is important in windy conditions by removing the warm insulating layer of air around the body. The preoptic nucleus of the anterior hypothalamus is the thermal control center, which maintains body temperature at a given set value. In response to a decrease in core body temperature, the hypothalamus initiates mechanisms to conserve heat by cutaneous vasoconstriction and produce heat by stimulation of muscular activity in the form of shivering.2,3 Nonshivering thermogenesis occurs via increased activity of thyroxine and catecholamines.4,5 In a conscious individual, the appreciation of cold induces the individual to exercise, wear more clothes, or move to a warmer environment.
As the core temperature decreases below 35°C (95°F), the coordinated systems responsible for thermoregulation begin to fail because the physiologic responses to minimize heat loss are very limited.6 Primary hypothermia (accidental hypothermia) refers to a spontaneous decrease of core body temperature, usually as result of exposure to cold environments without adequate protection. Environmental hypothermia results from a combination of heat loss by convection (degree of wind exposure), conduction, and radiation to the surrounding ambient air.Secondary hypothermia represents a complication of an underlying disorder. Some of the disorders and conditions that may predispose an individual to hypothermia by decreasing heat production, increasing heat loss, or interfering with the central or peripheral control of thermoregulation are listed in Table 131-1.1-3,6-10
Humans are capable of significant heat generation. Strenuous exercise can increase heat generation twentyfold.
Normally, the body dissipates heat into the environment via four mechanisms. The evaporation of skin moisture is the single most efficient mechanism of heat loss but becomes progressively ineffective as the relative humidity rises to >70%. The radiation of infrared electromagnetic energy directly into the surrounding environment occurs continuously. (Conversely, radiation is a major source of heat gain in hot climates.) Conduction— the direct transfer of heat to a cooler object—and convection—the loss of heat to air currents—become ineffective when the environmental temperature exceeds the skin temperature.
Factors that interfere with the evaporation of diaphoresis significantly increase the risk of heat illness. Examples include dripping of sweat off the skin, constrictive or occlusive clothing, dehydration, and excessive humidity. While air is an effective insulator, the thermal conductivity of water is 25 times greater than that of air at the same temperature. The wet-bulb globe temperature is a commonly used index to assess the environmental heat load. This calculation considers the ambient air temperature, the relative humidity, and the degree of radiant heat.
The regulation of this heat load is complex and involves the central nervous system (CNS), thermosensors, and thermoregulatory effectors. The central thermostat activates the effectors that produce peripheral vasodilation and sweating. The skin surface is in effect the radiator and the principal location of heat loss, since skin blood flow can increase 25–30 times over the basal rate. This dramatic increase in skin blood flow, coupled with the maintenance of peripheral vasodilation, efficiently radiates heat. At the same time, there is a compensatory vasoconstriction of the splanchnic and renal beds.
Acclimatization to heat reflects a constellation of physiologic adaptations that permit the body to lose heat more efficiently. This process often requires one to several weeks of exposure and work in a hot environment. During acclimatization, the thermoregulatory set point is altered, and this alteration affects the onset, volume, and content of diaphoresis. The threshold for the initiation of sweating is lowered, and the amount of sweat increases, with a lowered salt concentration. Sweating rates can be 1–2 L/h in acclimated individuals during heat stress. Plasma volume expansion also occurs and improves cutaneous vascular flow. The heart rate lowers, with a higher stroke volume. After the individual leaves the hot environment, improved tolerance to heat stress dissipates rapidly, the plasma volume decreases, and de-acclimatization occurs within weeks.
++A normal body temperature is ordinarily maintained despite environmental variations because the hypothalamic thermoregulatory center balances the excess heat production derived from metabolic activity in muscle and the liver with heat dissipation from the skin and lungs. According to studies of healthy individuals 18–40 years of age, the mean oral temperature is 36.8° ± 0.4°C (98.2° ± 0.7°F), with low levels at 6 a.m. and higher levels at 4–6 p.m. The maximal normal oral temperature is 37.2°C (98.9°F) at 6 a.m. and 37.7°C (99.9°F) at 4 p.m.; these values define the 99th percentile for healthy individuals. In light of these studies, an a.m. temperature of >37.2°C (>98.9°F) or a p.m. temperature of >37.7°C (>99.9°F) would define a fever. The normal daily temperature variation is typically 0.5°C (0.9°F). However, in some individuals recovering from a febrile illness, this daily variation can be as great as 1.0°C. During a febrile illness, the diurnal variation is usually maintained, but at higher, febrile levels. The daily temperature variation appears to be fixed in early childhood; in contrast, elderly individuals can exhibit a reduced ability to develop fever, with only a modest fever even in severe infections.
Rectal temperatures are generally 0.4°C (0.7°F) higher than oral readings. The lower oral readings are probably attributable to mouth breathing, which is a factor in patients with respiratory infections and rapid breathing. Lower-esophageal temperatures closely reflect core temperature. Tympanic membrane thermometers measure radiant heat from the tympanic membrane and nearby ear canal and display that absolute value (unadjusted mode) or a value automatically calculated from the absolute reading on the basis of nomograms relating the radiant temperature measured to actual core temperatures obtained in clinical studies (adjusted mode). These measurements, although convenient, may be more variable than directly determined oral or rectal values. Studies in adults show that readings are lower with unadjusted-mode than with adjusted-mode tympanic membrane thermometers and that unadjusted-mode tympanic membrane values are 0.8°C (1.6°F) lower than rectal temperatures.
In women who menstruate, the a.m. temperature is generally lower in the 2 weeks before ovulation; it then rises by ∼0.6°C (1°F) with ovulation and remains at that level until menses occur. Body temperature can be elevated in the postprandial state. Pregnancy and endocrinologic dysfunction also affect body temperature.
Content 6
{The hypothalamus integrates information from the environment, nervous system, and the other organ systems.}
3) Corticotrophin Releasing Hormone is released by the paraventricular nucleus and taken up by the portal system where it has its action on the anterior lobe of the pituitary. A recent, interesting development is the finding of a direct projection from the eye to the suprachiasmatic nucleus of the supraoptic hypothalamic region.
4) The median eminence—the bulge from which the infundibulum extends to the anterior pituitary . In the median eminence, a dense network of capillaries arises from the superior hypophyseal arteries. These capillaries drain into portal vessels that traverse the pituitary stalk and then form a capillary network within the anterior pituitary gland. The primary direction of this hypophyseal portal system is from hypothalamus to pituitary, however, retrograde flow also exists. This creates an ultrashort feedback loop between the pituitary gland and hypothalamic neurons.
5) The mammillary or posterior region at the level of the mammillary bodies [What does it secrete?]
6). Thermoregulation
sexually dimorphic nucleus (SDN, intermediate nucleus, INAH-1),
suprachiasmatic nucleus (SCN)
tuberal lateral nucleus (NTL).
The hypothalamus regulates body functions that vary at different times of the day (e.g., body temperature, hormone secretion, hunger) or those that vary over a period of many days (e.g., menstrual cycle). A projection from the retina to the suprachiasmatic nucleus is thought to supply the clock with day-night information needed for synchronizing diurnal (daily) rhythms (also known as circadian rhythms). Lesions of the hypothalamus often disrupt the state of the sleep-waking cycle.
This neural region is associated with hormonally related behaviour (anger and sexual activity), homeostatic regulation (blood pressure, heart rate, appetite, and temperature) and functions that relate to both (puberty, reproductive cycles, and lactation). The hypothalamus is a small part of the diencephalon that is involved
in the mediation of endocrine, autonomic and behavioral functions.book whenever the specific hormone systems to which they belong are described. Several neuropeptides have been isolated from the hypothalamus, and many continue to be discovered. However, only those that have been demonstrated to control anterior pituitary function (hypophysiotropic hormones) and, therefore, play an important role in endocrine physiology will be discussed.
GnRH is a small peptide produced in the hypothalamus by specialized nerve cells; as such, GnRH is called a neurohormone, a class of hormones that include thyrotropin-releasing hormone, oxytocin (see below), antidiuretic hormone (Chapter 18), and corticotropin-releasing hormone. Release of GnRH results in activation of a specific GnRH receptor, located in the gonadotropes of the pituitary gland. This receptor is a membrane-bound G-protein-coupled stimulator of phospholipase C, which results in calcium release and protein kinase C activation via conversion of plasma membrane phosphatidylinositol into inositol triphosphate and diacylglycerol (Chapter 8). These signals result in production and release of FSH and LH, as will be described below. Regulation of this important signal is multifold. GnRH is degraded within minutes so it is constantly produced in pulses, and the size and frequency of these pulses is important in signaling. These GnRH pulses are constant in males but vary in females, depending on the menstrual cycle. Interestingly, the frequency of the GnRH pulses result in different expression of FSH (low frequency) and LH (high frequency). As a result FSH and LH are variably expressed during the female menstrual period (Figure 20-2). Levels of testosterone, estrogen, and prolactin (increased during pregnancy) as well as increased concentration of FSH and LH create a negative feedback loop, which can decrease GnRH pulses.
(2) involved in temperature regulation, homeostatic regulation (blood pressure, heart rate, appetite, and temperature
(3) control of food and water intake (thirst),
4) sexual behavior and reproduction
(5) control of daily cycles in physiological state and behavior
(6) mediation of emotional responses,such as anger
(7) controls functions that relate to both (puberty, reproductive cycles, and lactation).
03. Hypothalamic–Pituitary–Adrenal Axis
04. Hypothalamic–Pituitary–Gonadal Axis
http://nba.uth.tmc.edu/neuroscience/s4/chapter01.html
- Chapter 1: Hypothalamus: Structural Organization, Patrick Dougherty, Ph.D.
- Chapter 2: Hypothalamic Control of Pituitary Hormones, Patrick Dougherty, Ph.D.
- Chapter 3: Central Control of the Autonomic Nervous System & Thermoregulation, Patrick Dougherty, Ph.D.
- Chapter 4: Central Control of Feeding Behavior, Patrick Dougherty, Ph.D.
- Chapter 5: Limbic System: Hippocampus, Anthony Wright, Ph.D.
- Chapter 6: Limbic System: Amygdala, Anthony Wright, Ph.D.
- Chapter 7: Learning and Memory, John H. Byrne, Ph.D.
- Chapter 8: Higher Cortical Functions: Language, Anthony Wright, Ph.D.
- Chapter 9: Higher Cortical Functions: Association and Executive Processing, Anthony Wright, Ph.D.
- Chapter 10: CNS Aging and Alzheimer's Disease, Jack Waymire, Ph.D.
- Chapter 11: Blood Brain Barrier and Cerebral Metabolism, Pramod Dash, Ph.D.
- Chapter 12: Neurotransmitter and Cell Death, Pramod Dash, Ph.D.
-
Oxytocin, and Mother-Baby Interactions
POSTED ON TUE, OCT 29, 2019 @ 01:00 PM BY LAURA SNIDER
The fact that it’s good for parents to spend quality time with their children is just common sense—but did you know that a new study showed that mothers’ behavior has an effect on the development of their babies’ oxytocin systems? A team of scientists from the University of Virginia and the Max Planck Institute for Human Cognitive and Brain Sciences found that “greater maternal involvement seems to have the potential to upregulate the oxytocin system in human offspring.” Before we go into exactly what that means, though, let’s talk about oxytocin for a second.
You may have heard of oxytocin before: people sometimes call it the “love hormone” or the “cuddle hormone.” It’s a versatile hormone, released in response to the activation of sensory nerves. During childbirth, it causes smooth muscle in the uterus to contract, and during breastfeeding, it stimulates milk ejection from the mammary glands. It’s released during sexual intercourse, skin-to-skin contact between mothers and infants, and positive human-human or human-animal social interactions (interactions between humans and dogs, for instance).
 The mammary glands (breast tissue) and uterus of the female reproductive system.
The mammary glands (breast tissue) and uterus of the female reproductive system.
Image from Human Anatomy Atlas.Want to know something else cool about oxytocin? It functions as both a hormone and a neurotransmitter.
Oxytocin is produced by the hypothalamus, and the posterior pituitary gland sends it off into systemic circulation to perform its childbirth and labor functions. Once oxytocin is out of the brain, it can’t get back in (due to the blood-brain barrier).
 Image from Human Anatomy Atlas.
Image from Human Anatomy Atlas. However, oxytocin acts within the brain as well, affecting the CNS. Oxytocinergic neurons (neurons that produce oxytocin as a neurotransmitter) project from the paraventricular nucleus (PVN) to important regulatory brain areas, which include “other nuclei within the hypothalamus, the amygdala, the hippocampus, the PAG, the frontal cortex, the raphe nuclei, the striatum, the [nucleus accumbens], the vagal nuclei (both the nucleus of the solitary tract (NTS) and the dorsal vagal nucleus (DMX), and the [locus coeruleus].”
The UVA/Max Planck study investigated whether babies had lower methylation of the oxytocin receptor gene (the gene that codes for a protein that serves as a receptor for oxytocin) when their mothers were more engaged during play. DNA methylation is an epigenetic process, meaning that it changes the expression of genes but doesn’t alter their sequence of nucleotides. DNA methylation usually represses gene expression.
Here’s how the study worked. Mothers and their five-month-old babies were recorded during a play session and rated on both maternal and infant factors. Maternal factors included how talkative the mother was, how close she was to the baby, how attentive the mother was to the baby’s needs, how positive the mother’s mood was, and the duration of passive and active touch. Infant factors included how attentive the baby was, how positive their mood was, and the duration of the baby’s smiles with and without vocalizations.
The mothers also filled out a breastfeeding questionnaire, a maternal demographics survey, a short form of the social support questionnaire (SSQ6), the Parenting Sense of Competence Scale, and the Edinburgh Postnatal Depression Scale. Keep in mind: this was an observation, not an experimental manipulation.
Saliva samples were collected when the children were five months old (this was when the play sessions were recorded as well), and then around a year later, when the children were 18 months old.
Ultimately, the study found that there was less methylation of the oxytocin receptor gene at 18 months in mother-baby pairs where the mother was more engaged during play at 5 months. The mothers’ levels of OXTR methylation did not change significantly between the 5- and 18-month samples. On the basis of these results, the authors suggest that “infancy may provide a sensitive period in development during which the oxytocin system is dynamic, plastic, and sensitive to the social environment.”
Reference
https://human-memory.net/hypothalamus/
- Identify the two hormones released from the posterior pituitary, their target cells, and their principal actions
- Identify the six hormones produced by the anterior lobe of the pituitary gland, their target cells, their principal actions, and their regulation by the hypothalamus